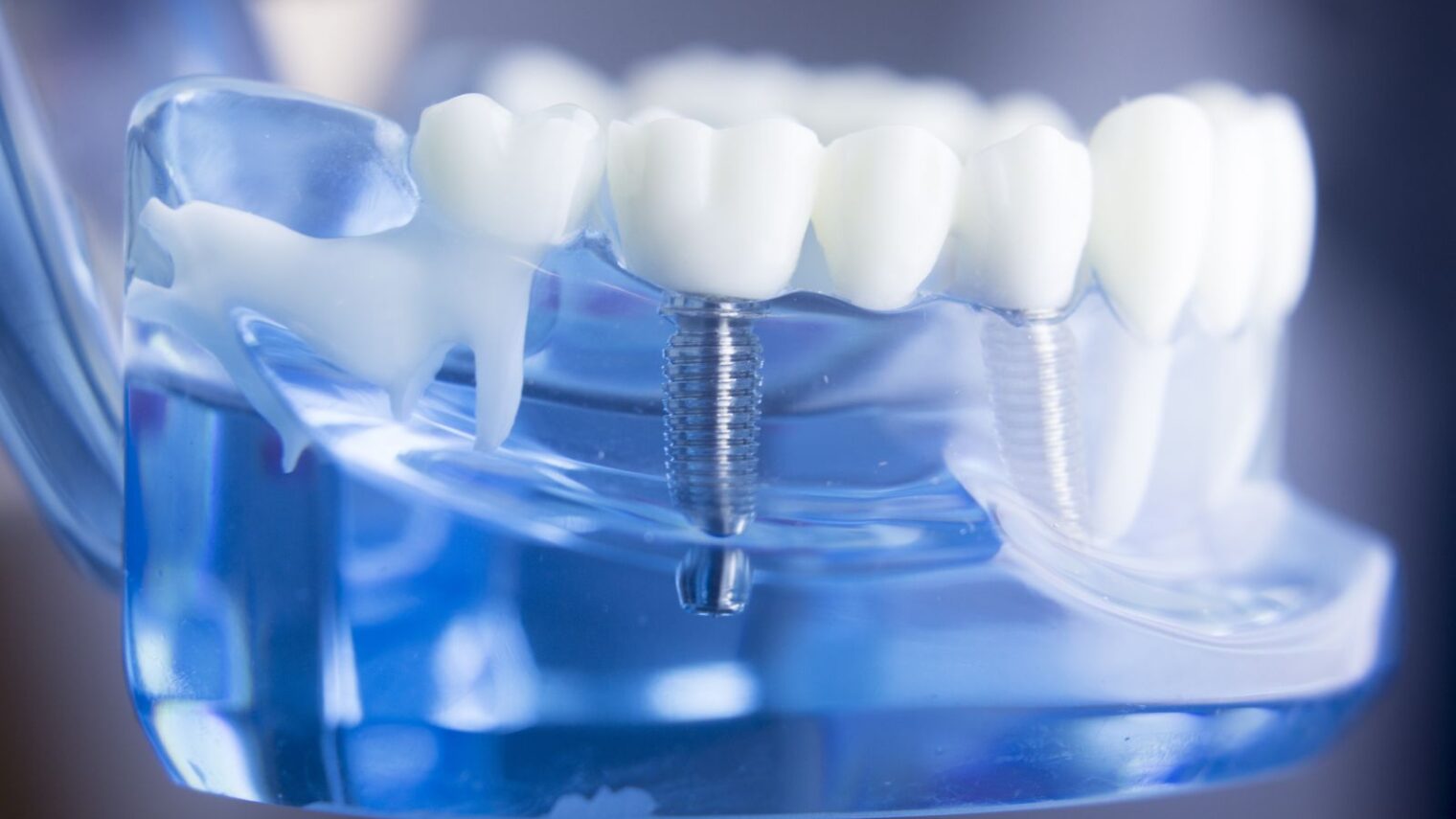
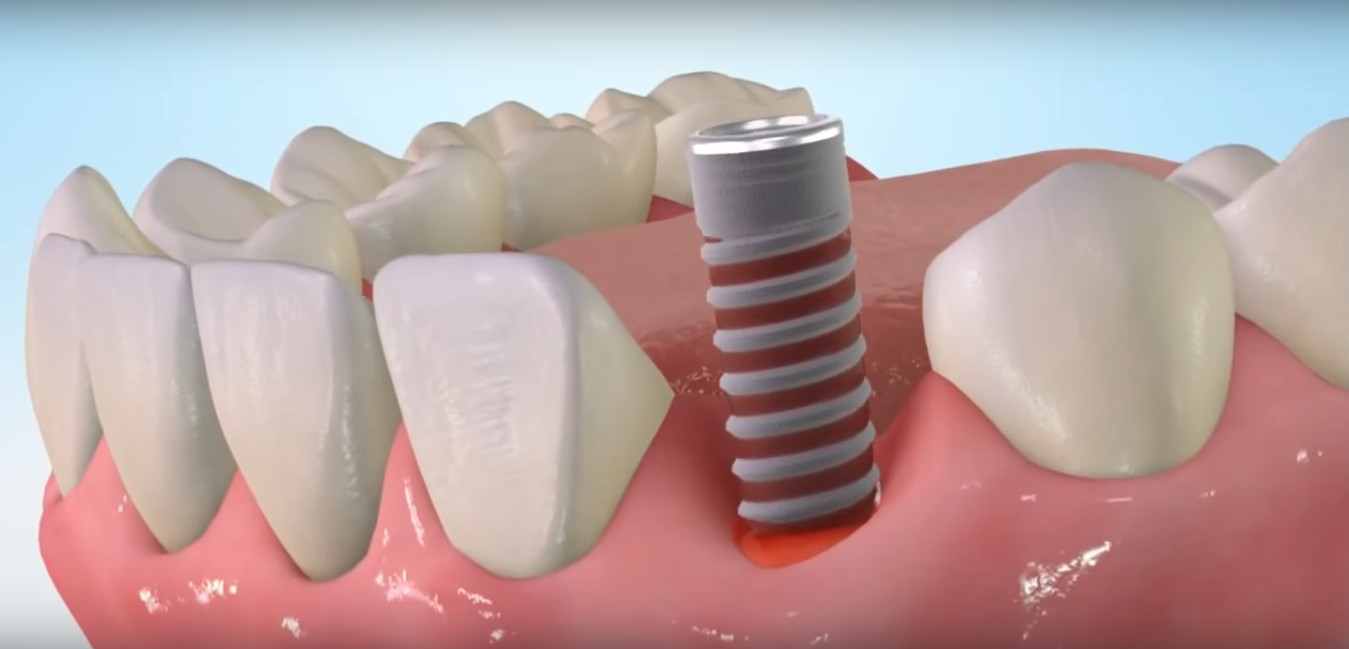
A unique cold-plasma activation platform from Israeli company Nova Plasma is poised to hasten healing and reduce infection after the placement of all kinds of surgical implants, from dental to breast.
The first application of the company’s Active+ Plasma Capsule is a desktop device that modifies the surface chemistry of dental implants in a 30-second procedure right before implantation, for maximum effectiveness.
Plasma, a partially ionized gas, has only recently been discovered to have vast potential in medical settings to weld tissue, control bleeding, enhance tissue repair, disinfect and even destroy cancer cells.
Nova Plasma was cofounded in 2012 by two Israeli brothers, CEO Ronen Lam and CTO Amnon Lam, along with Chen Porat. The Lams pioneered the activation of cold plasma at their previous company, IonMed, which is focused on sealing incisions.
They remain shareholders in IonMed but the two companies do not share any intellectual property on their very different technologies.
Ronen Lam tells ISRAEL21c that Nova Plasma originally intended to develop a product for tooth-whitening. And then a commercial company told them of the unmet need for improving the surface of dental implants.
“Many articles and trials have shown plasma’s amazing effect on the surface of implants but there was no way to do this practically because the plasma effect on the surface is limited in time and thus not possible to be performed in the factory, so they asked us to build a ‘chairside’ machine designed to be operated in the dental clinic. We did feasibility studies and saw that our cold plasma does the job in a perfect way. Then we came up with a very sophisticated idea for applying the plasma in the clinic, and all the IP went in this direction.”
Nova Plasma signed a licensing agreement with a leading player in the dental market, and is moving from development to production in preparation for introducing Active+ later this year.

Nova Plasma’s cold-plasma platform technology could be applied to many other implants, including pacemakers, artificial joints and catheters.
Lam explains that the integration and stability of all implants are improved by their wettability (hydrophilicity). Plasma is great for activating hydrophilicity, but the effects deteriorate in the time lapse between manufacturing and implantation.
Nova’s Active+ Plasma Capsule solves that problem by activating the plasma surface treatment immediately before implantation. The resulting “super hydrophilic” implant reduces average healing time by half, improves implant anchoring in the jaw, and reduces post-surgical complication rates. No training is required for operating the machine.
“It’s really easy and intuitive,” says Lam.
A $1.5 million financing round has just been launched to advance Nova Plasma’s next product, Active Si, for modifying the surface of silicone breast implants before they are dipped in antibiotic solution prior to implantation. Pre-clinical studies show that this method eliminates biofilm formation on silicone.
“This product is at the end of development, preparing for clinical trials starting in October,” says Lam. “The need is huge in the field of breast implants because there are complications in up to 30 percent of cases. In addition, the aesthetic results are expected to be improved using Active-Si technology. We’ll start taking it to market in a controlled manner in a few leading medical centers in Europe. We believe it will be at a fully commercial stage in 2019.”
Based in Kibbutz Megiddo, Nova Plasma has until now been financed by current shareholders, including many of its medical and IP advisers and entrepreneurs.
“One of the biggest assets of our company, besides a strong and beautiful platform technology with huge potential, is our development team’s knowledge. I can say without exaggeration that it may be one of the world’s best teams in bringing cold plasma to market, and applying it through regulatory obstacles while meeting the business model,” says Lam.
For more information, click here.