Whether you call them bedsores, pressure ulcers or pressure injuries (PI), they’re a serious healthcare risk to people confined to a bed or chair and to people with diabetes and poor circulation.
The red, painful sores often affect heels, ankles, hips and tailbone. They can become infected and extend into the muscle and bone if not caught and treated quickly. They can even be deadly.
However, the current gold standard for PI monitoring is a twice-weekly visual check of at-risk hospitalized patients.
Not only does this method fail to catch PIs at their earliest stages – they can develop within hours – but it often fails to detect PIs on dark skin.
This problem has become a major healthcare equity issue in the United States, where later-stage PIs are more prevalent in people with darker skin tones.

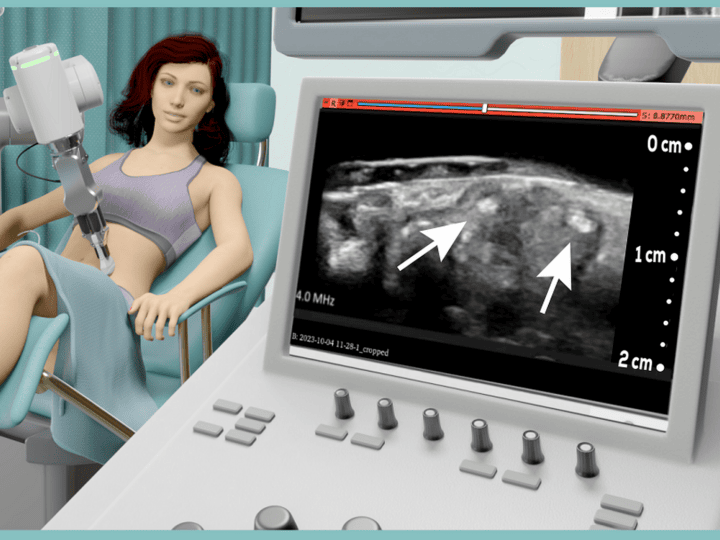
Israel-based IR-Med plans to revolutionize PI detection and decision-making with a device powered by infrared (IR) light spectroscopy and artificial intelligence (AI).
The PressureSafe device noninvasively checks under the patient’s skin for biomarkers of pressure injuries forming before they are outwardly visible.
“There is high demand for objective devices that further healthcare equity,” says IR-MED cofounder and Chief Scientific Officer Yaniv Cohen, who has a PhD in biotech engineering.
“Our device works similarly in terms of effectiveness in people with light and dark skin. That’s the beauty of infrared light.”
Recently, IR-Med presented its technology at the annual conference of the National Pressure Injury Advisory Panel in San Diego at a workshop on detecting early skin changes in dark skin tones, and also at Northwell Health’s 10th Annual Shining the Light on Wound Care Symposium in New York.
US hospitals
Founded in 2017 by Cohen and biochemical engineer Ronnie Klein, IR-Med aims to fill an unmet need in a worldwide PI treatment market valued at $4.5 billion in 2021 and expected to reach $6.4 billion by 2027.

In the United States alone, PIs affect an estimated 1 to 3 million people each year. The national cost of hospital-acquired pressure injuries alone is about $26.8 billion.
“Our founders researched infrared technology as it relates to identifying, measuring and monitoring different situations in body tissues in a noninvasive manner,” says Cohen.
Hospital testing revealed that infrared light in specific spectrums, when analyzed with AI, is highly accurate in identifying certain types of injuries under the skin before they evolve into an open wound.
As a platform technology, it’s also a potentially game-changing way to monitor drug levels in the blood in a noninvasive manner, as was proven in IR-MED’s study monitoring levels of anesthetic drugs in the bloodstream during surgery.
IR-Med, which went public in 2021, chose PI detection as its first go-to-market product. Cohen expects to submit PressureSafe for registration with the US Food and Drug Administration and begin sales before the end of the year.
“We are in discussions with a few chains of hospitals and nursing homes in the United States, which will be our initial customers as soon as we are ready,” says Cohen.
Beta testing
The company’s alpha device is undergoing studies at two Israeli hospitals to assess how it compares to the current visual assessment method.
Beit Rivka Medical Center in Petah Tikva, a 291-bed geriatric facility for skilled nursing, intensive care and rehab, is one of the testing sites.
The principal investigator is geriatric and internal medicine specialist Dr. Gal Maidan, Beit Rivka’s deputy general manager and head of innovation.
“We see a tremendous unmet need in this area. The use of infrared light to detect the early stages of pressure wounds and tissue damage has the potential to fill the gaps where other methods fail,” Maidan tells ISRAEL21c.
“It can serve almost as a preventive measure since intervention when the skin is still intact can more easily change the course of the tissue injury.”
The trial will continue another three months. “Our aim is to finish this study soon and publish results before the end of this year,” Maidan says.
Cohen reports “very good and accurate results” thus far. Meanwhile, IR-Med is
manufacturing the commercial model of PressureSafe, ergonomically designed by an industrial engineer.

The next step is to adapt the same device to detect foot wounds in people with diabetes.
“Usually they are checked every six months for these wounds, and that test is also visual so it suffers from the same deficiencies as the pressure injury test,” Cohen says. “We are in discussion with a hospital in Israel to study this indication.”
Also in IR-Med’s pipeline are a product to check noninvasively the level of various drugs in the blood, as well as a device using infrared light to determine whether children’s ear infections are caused by a virus or bacteria.
“Doctors usually prescribe antibiotics because there is no way to know. With our device, doctor will have the information to decide whether an antibiotic is appropriate, especially for hard-to-diagnose middle-ear infections.”
For more information, click here.