Mumbai-based Sun Pharmaceutical Industries has signed an exclusive worldwide licensing deal to further develop MM-II, a novel pharmaceutical candidate for treating pain in osteoarthritis, developed by Tel Aviv’s Moebius Medical.
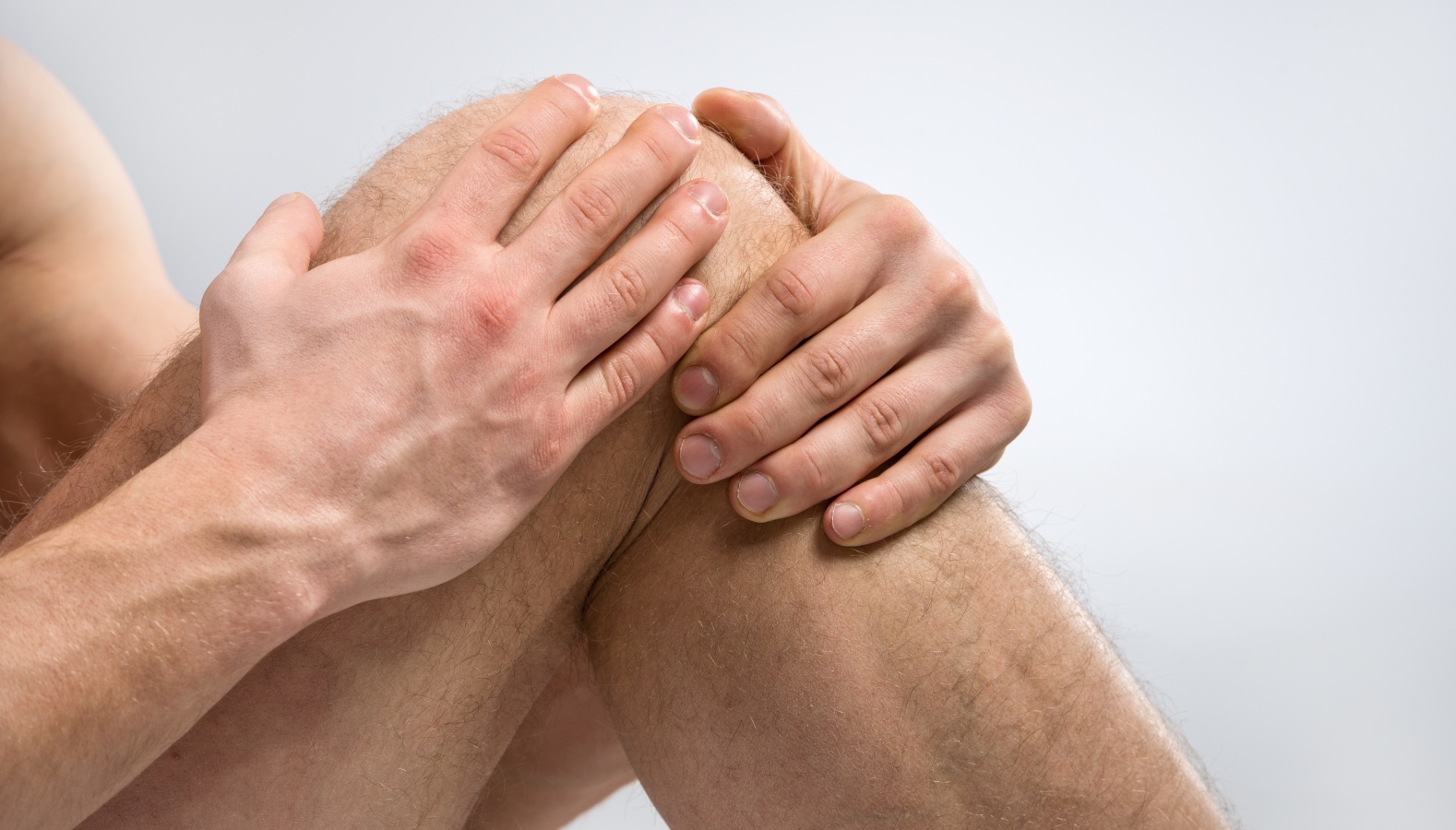
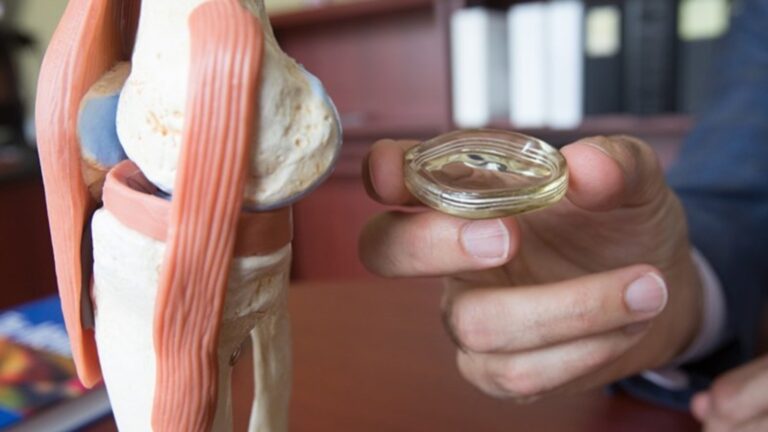
MM-II is a novel non-opioid product that leverages the physical properties of proprietary liposomes to lubricate arthritic knee joints, thereby reducing friction and wear and consequently reducing joint pain.
The product is based on patent-protected technology licensed by Moebius Medical from the tech-transfer companies of Hebrew University of Jerusalem, Technion-Israel Institute of Technology and Hadassah University Medical Center.
The bio-lubricant injection is intended to provide symptomatic relief of mild-to-moderate osteoarthritis pain. Osteoarthritis is one of the most common chronic health conditions and a leading cause of pain and disability among adults, affecting more than 100 million people. More than 20 million US citizens suffer from knee osteoarthritis.
“The fact that our novel technology was conceived in Israel and developed within the RAD Biomed Accelerator, confirms the unique quality of the country’s biotechnology ecosystem,” said Moshe Weinstein, CEO of Moebius Medical.
“Our technology was born from the multidisciplinary cooperation between leading professors from three of Israel’s most prestigious research institutions: Prof. Yechezkel Barenholz of the Hebrew University, Prof. Izhak Etsion of the Technion Institute, and Prof. Dorit Nitzan of Hadassah Medical Center.”
Sun Pharma, the world’s fifth largest specialty generic pharmaceutical company and India’s top pharmaceutical company, will fund further development of MM-II and undertake its global commercialization.
Moebius Medical has completed a first-in-man clinical study at Hadassah Medical Center, demonstrating the product’s fast onset of action and its potentially better efficacy and comparable safety for alleviating osteoarthritis pain as compared to hyaluronic acid injection.
Fighting for Israel's truth
We cover what makes life in Israel so special — it's people. A non-profit organization, ISRAEL21c's team of journalists are committed to telling stories that humanize Israelis and show their positive impact on our world. You can bring these stories to life by making a donation of $6/month.