For patients suffering from heart valve or aortic insufficiency, the only solution currently available is open-heart surgery to replace the valve.
Unfortunately, the five- to six-hour operation is too dangerous for some people, especially older adults. Only 20 percent of patients can be approved for the procedure.
Yet, if left untreated, heart valve insufficiency can lead to heart palpitations, chest pain, fatigue, shortness of breath and, ultimately, heart failure. The five-year survival rate is just 30%.
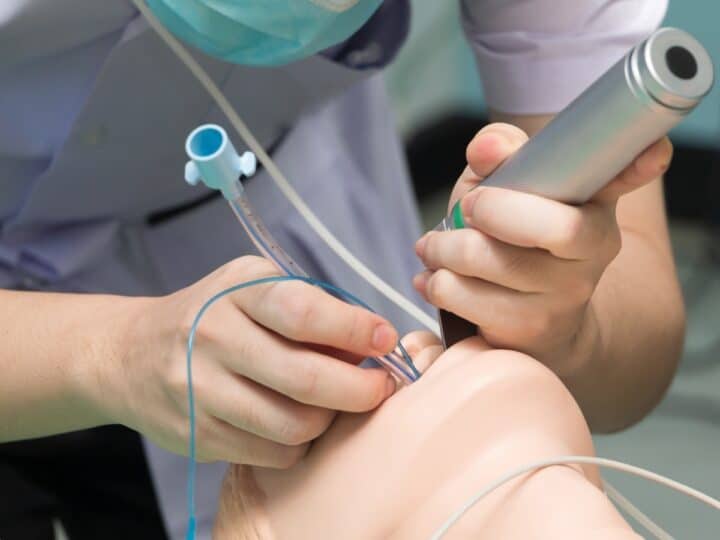
Israeli medical device startup Cuspa Medical is aiming to treat heart valve insufficiency with a procedure similar to how a stent is inserted into the heart – via a catheter threaded through an artery that starts in the groin area.
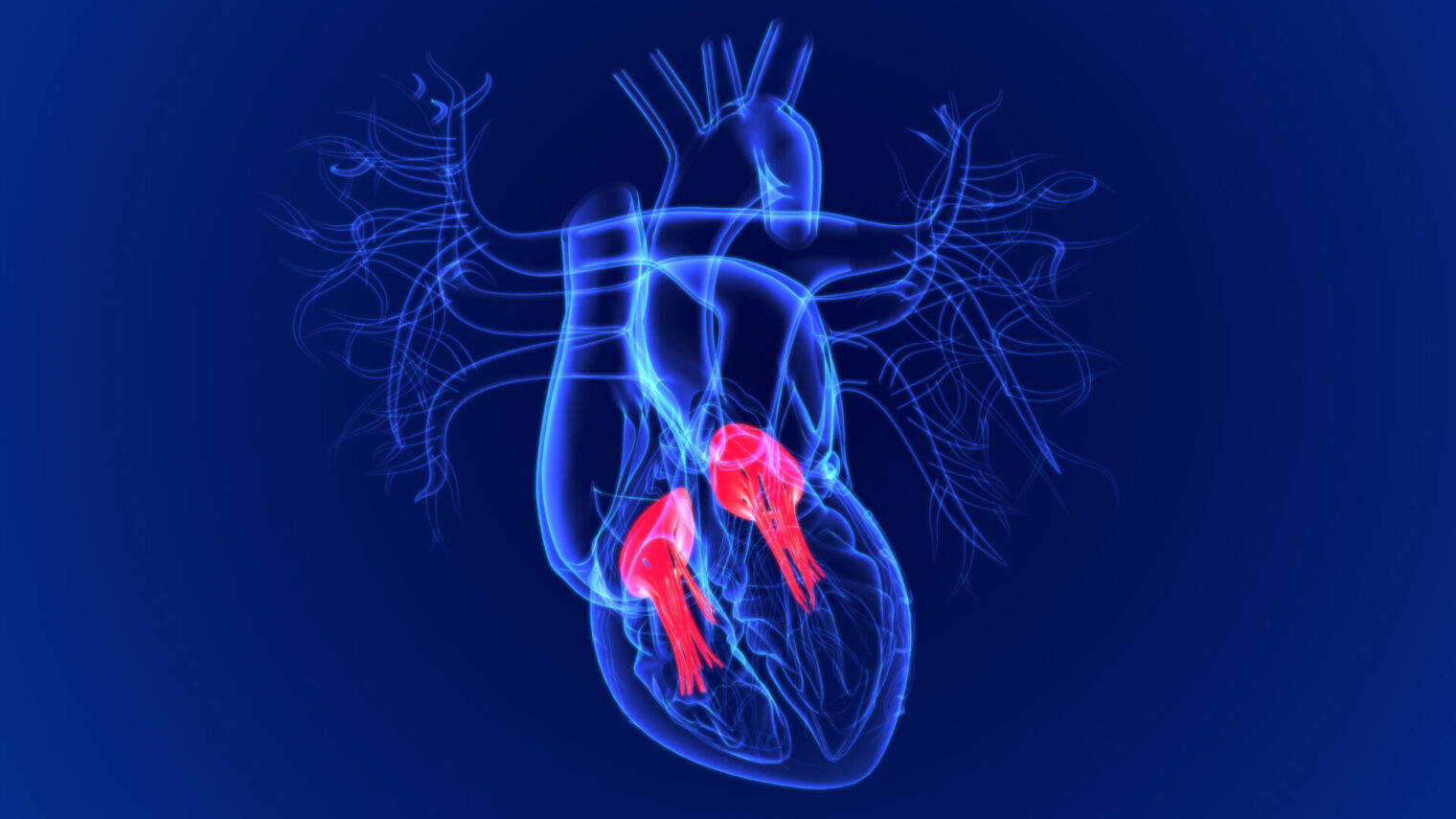
As Cuspa’s minimally invasive Cusper device arrives at the heart, it “grasps” the leaflets (strong flaps of tissue) around the aorta, sealing them to prevent the leakage of blood through the heart valve.
Leakage is why heart valve insufficiency is so dangerous.
The aortic valve sits between the left ventricle of the heart and the aorta. If the valve doesn’t close well, instead of blood flowing from the ventricle to the aorta, it flows back to the left ventricle and creates stress on the heart.
The condition is known as “aortic regurgitation” and is reminiscent of GERD (gastro-esophageal reflux disease) where the lower esophageal sphincter becomes weak, leading to digestive acid being regurgitated and causing persistent heartburn.
But while GERD can be a pain, aortic regurgitation can be deadly.
Testing on pigs
Cuspa has so far tested Cusper on healthy pigs.
“We’re mainly testing the safety of the device at this point,” Ariel Weigler, Cuspa’s CEO, tells ISRAEL21c.
When aortic regurgitation is “created” in the test pigs, Cusper reduces regurgitation by up to 80% while staying in place and not generating any tension or other damage to the leaflets.
If it works in humans as well, Cusper “has the potential to move patients from a severe to mild case, regaining quality of life. This will be verified in the first in human trials,” Weigler explains.

Cuspa’s clamp is made of the same kind of metal alloy used for stents – nitinol – which does not promote any reaction from the body’s tissues and “remembers” the shape it’s supposed to assume.
“It can crimp into a very small size,” Weigler says, making it possible to move from the groin to the heart. After reaching the desired position through the catheter, the implant can be maneuvered into place.
“Once you release [nitinol], it reverts to a normal shape. That’s why it’s so widely used in all kinds of medical devices,” Weigler notes.
Because positioning Cusper in the body is so similar to inserting a stent, “physicians are very used to this procedure. There’s nothing holding them back from doing it,” Weigler says.
Inserting a Cusper is currently a hospital procedure, but Weigler hopes that in the future it could be done in an outpatient clinic.
The clamp remains on one of the aortic leaflets, and the patient could go home the same day.
Valve repair vs. replacement
Cuspa intends to price its product at about $20,000, including the device, the catheter and training.
Cuspa is not the only company trying to crack the problem of aortic insufficiency with a minimally invasive device.
California-based JenaValve, which raised $100 million earlier this year, is running tests in Europe, which represents about a third of the global market for aortic valve replacement. While JenaValve also uses the same artery to get from groin to heart, the product replaces the valve, rather than repair it, as Cuspa proposes.
Why not replace the valve like JenaValve? we asked Weigler.
“In most cases, the valve tissue is not the issue. The leaflets are still intact and operate normally while the insufficiency is caused by structural changes of areas around the valve itself. Preserving the native tissue is beneficial to the patient so it makes sense to repair the valve rather than replace it.”
Valve replacement also comes with side effects and complications, including the need to implant a pacemaker.
Raising $2 million
The problem Cuspa has set out to address affects about 10 million patients worldwide. The market was worth about $11 billion in 2021 and is expected to grow to $33 billion by 2030, driven in large part by an aging population.
Cuspa began life in Nazareth in the NGT3 incubator dedicated to fostering Jewish/Arab high-tech collaboration in medical devices and life sciences.
The company raised money through the incubator and the Israel Innovation Authority; Weigler and team are now looking to raise $2 million through a crowdfunding campaign. The goal is to finance another 18 months of testing on pigs before the human testing phase.
Cuspa’s team of four includes the company’s founder and chief medical officer, serial entrepreneur Dr. Yair Feld, who invented the technology.
If the clinical trials go according to plan, Weigler says, the device and treatment protocol could be commercialized within a few years. Testing is currently underway at Shamir Medical Center, along with additional tests on animals in the Beersheva area.
For more information, click here.