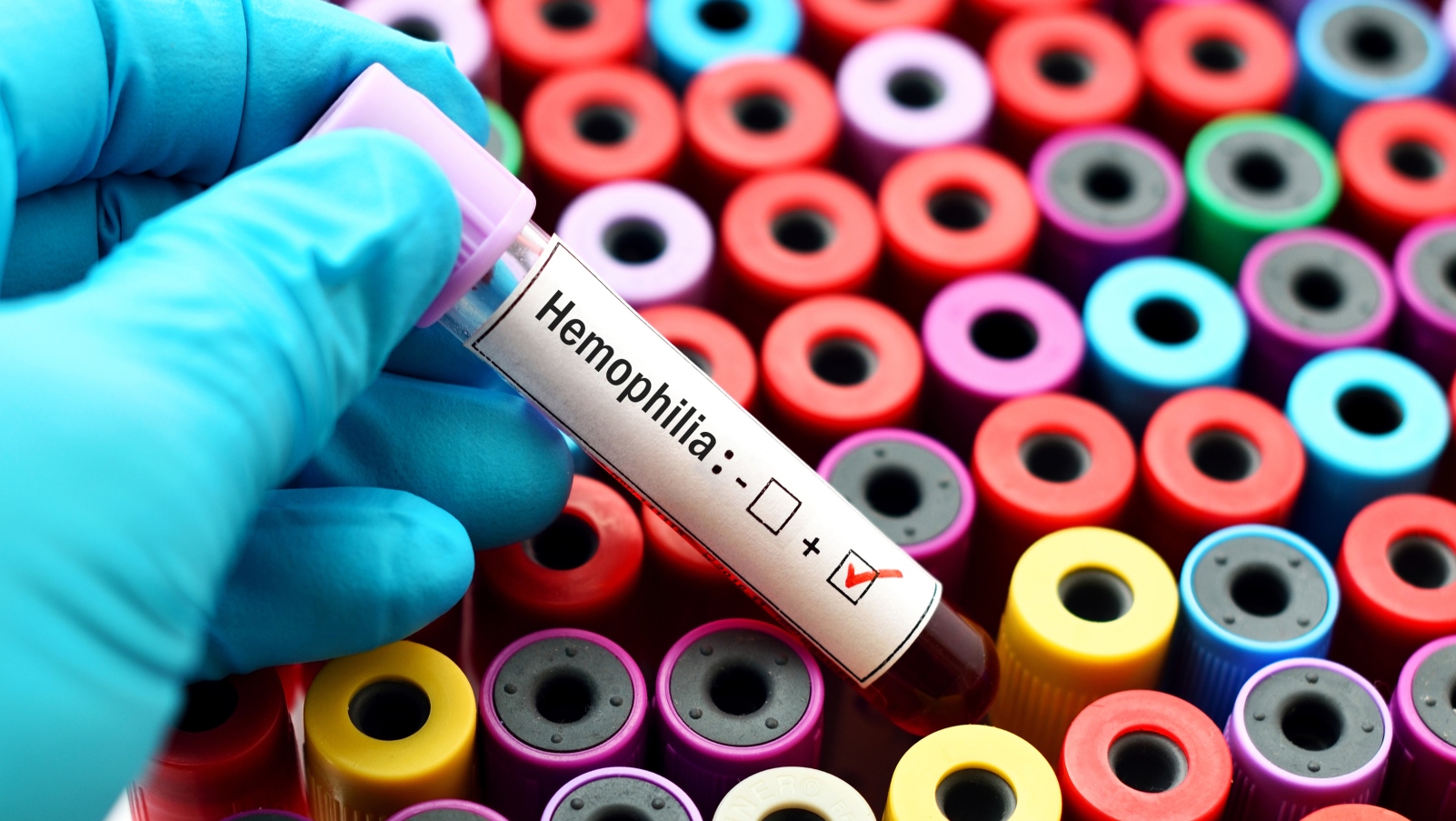
Is a cure for hemophilia on the horizon? American biotechnology company BioMarin aims to find out with the help of Sheba Medical Center in Ramat Gan, Israel.
BioMarin has chosen Sheba to take part in the next phase of international trials for what’s being touted as a “magic bullet” cure for hemophilia A, a genetic blood-clotting deficiency that results in patients bleeding longer after an injury and more easily bruising. Hemophilia A, which affects mostly men, can be life-threatening.
BioMarin has developed a treatment using viruses to implant new genes into a patient’s cells; a single dose seems to be enough to stop or even cure the disease.
BioMarin has conducted an initial trial on nine patients – all stopped using clotting factor drugs to treat their hemophilia and their annualized bleeding rate fell from some 16 episodes a year to just one.
Prof. Gili Kenet will run the project in Israel. Kenet, the director of the National Hemophilia Center and Institute of Thrombosis and Hemostasis at Sheba Medical Center, is impressed by what she’s seen so far.
“People in the medical community are beginning to speak about curing hemophilia after a one-time treatment with this new drug,” she said.
Sheba’s National Hemophilia Center treats more than 700 hemophiliacs a year and has about 50 patients currently participating in clinical studies. Kenet, an internationally known expert on hemophilia, thrombosis and hemostasis, is the current co-chair of the scientific subcommittee dealing with hemophilia A for the International Society of Thrombosis and Hemostasis.
“These are exciting times, as there are options to cure hemophiliac patients,” Kenet said. “We are hoping that results of this trial at Sheba will be good enough that this will be the therapy of choice that will able to stop this dreaded disease once and for all.”
Pending approval by the Israeli Ministry of Health, Sheba expects to begin enrolling patients shortly.