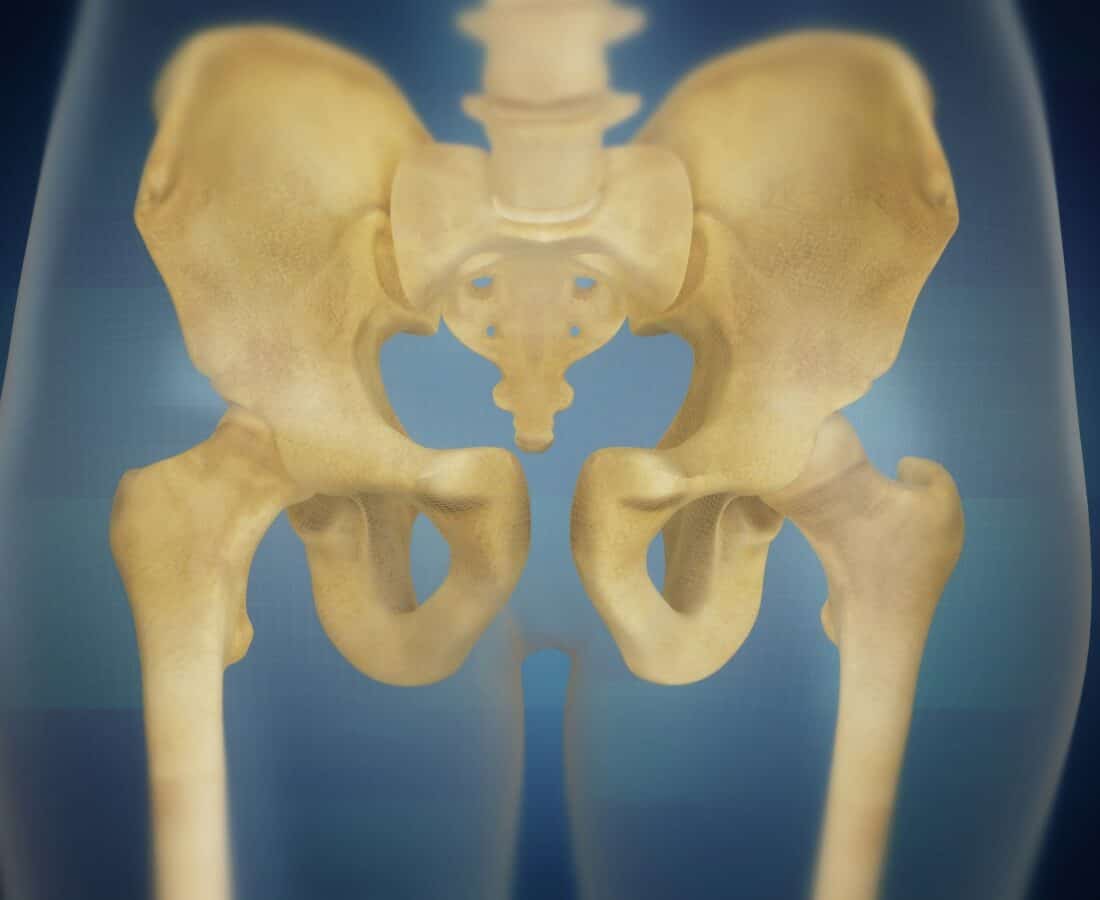
The pelvic floor is a “hammock” of muscles and connective tissues holding up a woman’s uterus, bowel, bladder, and vagina.
This hammock often weakens, resulting in pelvic floor disorders such as pelvic organ prolapse (POP), affecting up to half of all women over age 30.
Israeli OB/GYN and urogynecologist Dr. Elan Ziv says that less than 10 percent of affected women ask their doctor about POP, and only 2.9% are treated.
Still, the numbers are huge. In the United States alone, some 1.4 million women are fitted with supportive pessary devices each year and another 200,000 undergo POP repair surgery.
Stating the obvious, Ziv says women aren’t wild about either of those solutions.
He’s offering a different approach through his 20-year-old medical device company, ConTIPI Medical.
Below we’ll also look at how two other Israeli femtech companies have introduced minimally invasive surgical alternatives to address this medical problem affecting millions of women worldwide.
ProVate
Caesarea-based ConTIPI is introducing a unique disposable pessary that a woman can insert and remove at home. The physician only needs to write the prescription.
The FDA and CE cleared ProVate is very similar to a tampon so it’s easy for women to use. The applicator holds a supportive ring attached to a string. Tugging on the string collapses the ring for comfortable removal, which is done about every seven days.
“Nothing in the market even resembles this,” says Ziv.
Impressa, his previous invention for home management of female stress urinary incontinence, was designed along the same lines and was acquired by Kimberly Clark in 2013.
“The point of all our devices is to be somewhere between a menstrual tampon and a medical device,” Ziv tells ISRAEL21c.
“They come in small sizes with an applicator, made for shifting control to the user herself with no necessity for medical insertion. We only do noninvasive and disposable solutions for home use.”
He emphasizes that some 80% of POP cases can be managed non-surgically. But women don’t like pessaries, he says, which must be removed periodically for cleaning and reinsertion, usually by the doctor. ProVate is intended as a more user-friendly alternative.
ConTIPI recently raised $40 million from Capital IP Investment Partners and Ghost Tree Partners to bring ProVate to the US market next year through life-science commercialization company Indegene.
POP repair
Like any pessary solution, ProVate manages but does not repair POP.
For women seeking a more permanent solution – but without open or laparoscopic surgery requiring vaginal dissection — Israel’s Escala Medical and FEMSelect (formerly POP Medical Solutions) bring minimally invasive options to the field. Both are FDA and CE cleared.
Escala’s device, Apyx, anchors sutures into ligaments on one or both sides of the pelvic floor in a procedure that can be performed vaginally under local anesthesia in five to eight minutes in a doctor’s office.
“It mimics the gold-standard surgical repair procedure for all stages of prolapse. It’s the only solution that is entirely incision-free,” CEO Dr. Edit Goldberg tells ISRAEL21c.
The device also has a unique retriever that allows the procedure to be reversed if necessary.
Escala, a portfolio company of The Trendlines Group, recently received a total of 5.5 million euros in grant and equity investment from the European Innovation Council.
FEMSelect’s EnPlace device attaches anchors and sutures to ligaments of the pelvic floor at the sides and top, requiring only a small incision to anchor to the cervix.
Co-CEO Debbie Garner likens the procedure to putting up a tent with two “stakes” holding up the “roof.”

Designed by urogynecologist Dr. Menahem Neuman and engineer Boaz Harari, EnPlace includes a unique finger-guide delivery system to ensure proper placement.
The procedure can be used on women with mild, moderate or severe prolapse, and takes approximately 30 minutes under sedation in a day surgery center.
A recently published four-year follow up evaluation found that EnPlace had a success rate of 92.3%.
EnPlace is sold currently in Israel and in the United States, where FEMSelect and LiNA Medical USA handle promotion, distribution and physician education.
Because millions of women and their doctors are eager for new ways to deal with POP, Garner points out that there’s plenty of room for a variety of similar products.
“More solutions are better for everyone,” she says.