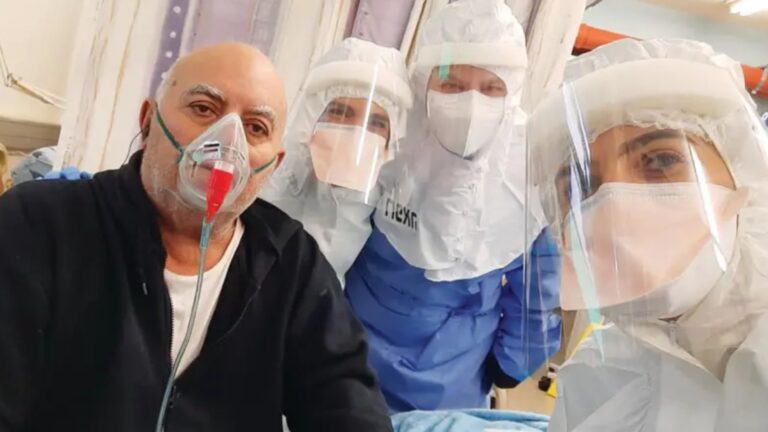
Ebola virus disease is a rare but severe illness that is fatal in 50 percent of cases, and those who survive may have symptoms for two years or longer. Patients must be hospitalized and receive oral or intravenous fluids and medicines including monoclonal antibody treatments.
Opaganib, a novel, twice-daily, oral medication developed by Israel’s RedHill Biopharma, delivered a statistically significant increase in survival time in a United States Army Medical Research Institute of Infectious Diseases (USAMRIID) in vivo Ebola virus study.
Opaganib is believed to be the first host-directed molecule to show activity in Ebola virus disease, having previously shown in vitro benefit in several strains of Ebola virus disease models.
“Given the unmet medical need and the untapped potential of host-directed antivirals, these results with opaganib, an easy to distribute and administer oral small molecule drug, support its further investigation for use in treating Ebola,” said study leader Rekha Panchal of USAMRIID.
Reza Fathi, RedHill’s senior vice president of R&D, said, “Opaganib has shown its host-directed antiviral potential in clinical and non-clinical studies, warranting further investigation in Ebola and other infectious viral diseases.”
Twice daily administered opaganib has previously demonstrated antiviral benefit in late-stage clinical studies of patients hospitalized with moderate to severe COVID-19. The drug was also selected by the National Institutes of Health Radiation and Nuclear Countermeasures Program for acute radiation syndrome.
Opaganib works through the inhibition of multiple pathways. It has anti-inflammatory properties and disrupts viral replication.
“We believe opaganib offers a potential breakthrough for fighting a virus capable of causing devastating outbreaks of disease in the countries least equipped to cope with them,” said Fathi.
Fighting for Israel's truth
We cover what makes life in Israel so special — it's people. A non-profit organization, ISRAEL21c's team of journalists are committed to telling stories that humanize Israelis and show their positive impact on our world. You can bring these stories to life by making a donation of $6/month.