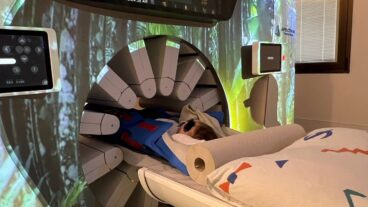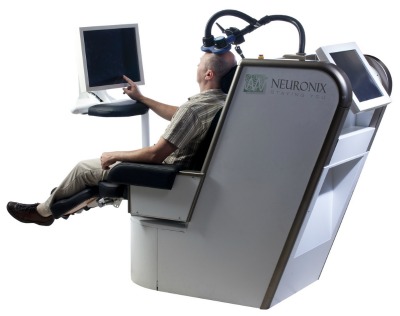
A new electromagnetic stimulation system has been developed in Israel that appears to change the course of the degenerative Alzheimer’s disease and allow patients to regain faded cognitive skills.
The non-invasive NeuroAD system, developed by Yokneam-based Neuronix, is the first medical device in the world to receive approval for treating mild to moderate Alzheimer’s disease, a terminal brain disease affecting about 30 million elderly people worldwide.
Scientific advisory board member Prof. José (Martin) Rabey, chief of neurology at Assaf Harofeh Medical Center in Tzrifin, unveiled NeuroAD in July at the annual International Conference on Alzheimer’s Disease in Paris.
The company’s solution is based on a patent-pending technology dubbed NICE (Non-Invasive Cortical Enhancer), which electromagnetically stimulates areas of the brain responsible for memory and learning, making them receptive to simultaneous tailored cognitive training. This double-barreled approach is hoped to induce LTP (long-term potentiation) associated with learning and memory processes.
‘Nothing else is available’
Clinical results show measurable cognitive improvement after a few weeks of treatment that is superior to improvements achieved with available drugs, says Neuronix CEO Eyal Baror, and NeuroAD can be used in conjunction with medication.
Even one year later, trial subjects maintained improvement in cognition, activities of daily living and decision-making to a state comparable to two years before treatment.
“The idea comes from an understanding of how Alzheimer’s surrounds cortical regions of the brain, moving from one area to another,” Baror tells ISRAEL21c. “We identified some regions more responsible for decline, and then came the idea for enhancing LTP, which is responsible for memory and learning, and degrades in Alzheimer’s disease.”
Baror reports that the concept is well-received in the worldwide medical community. “Wherever we go, physicians are eager to hear about this new technology because nothing else is available to help patients,” he says.
The initial two trials took place at Assaf Harofeh, resulting in “statistically significant results.” In May, new studies began under the supervision of Neuronix scientific advisers at Assaf Harofeh and at Boston’s Beth Israel Deaconess Medical Center, affiliated with Harvard Medical School.
Starting trials in the US next year
“We hope to open one or two more sites in Europe this year,” says Neuronix VP marketing Orly Bar. Following meetings with the US Food and Drug Administration later this year, the company also expects to expand its American clinical testing sites to 15 during 2012.
Trial participants undergo an MRI scan of the brain to pinpoint affected regions, and then receive five one-hour sessions per week for six weeks, in addition to long-term maintenance and follow-up. The treatment is painless and non-invasive, and can be repeated after its effects begin to wane, Bar adds.
The company was founded in 2008 by Dr. Jonathan Bentwich, who invented the platform technology and used it successfully to treat his mother, a former physician and Alzheimer’s patient. Bentwich, who left Neuronix in 2009, is the lead author in a 2011 article in the Journal of Neural Transmission about the efficacy of the device. Neuronix currently has 12 employees, half of them engaged in the engineering side of the enterprise. Funding comes from private investors and the office of Israel’s Chief Scientist.
Baror cannot yet predict the pricing of the made-in-Israel machine, but “our goal is to make it very affordable for all patients,” he says.
A graduate of the Israel Defense Forces prestigious Talpiot program, which allows promising young scientists and mathematicians to pursue higher education to further military research and development, Baror came to Neuronix with management experience at other medical device companies.
“LTP is known to degrade in patients with Alzheimer’s disease and can be re-stimulated by high frequency electro-magnetic pulses,” he says. With LTP reactivated, targeted cognitive training can be delivered with an enhanced and long-lasting effect, temporarily reversing the progress of the disease.”