Israeli scientists recently made history when they built a bionic liver and it revealed new insights about toxicity in a common pain reliever. This invention could be the solution sought by both scientists and animal-welfare activists seeking to put an end to live animal testing.
Due to consumer demands for “cruelty-free” products, new regulations (especially in Europe) are restricting the use of live animals in product safety tests. And scientists are increasingly concerned that animal studies do not accurately predict the human response to a beauty product or pharmaceutical, especially one that is used on a regular basis.
Using human cells for safety testing is not practical because the cells die after a few days outside the body. To address this challenge, scientists at the Hebrew University of Jerusalem and the Fraunhofer Institute for Cell Therapy and Immunology in Germany have created a liver-on-chip device mimicking human physiology.
They call it a 3D micro-reactor, or in less scientific terminology, a bionic liver.
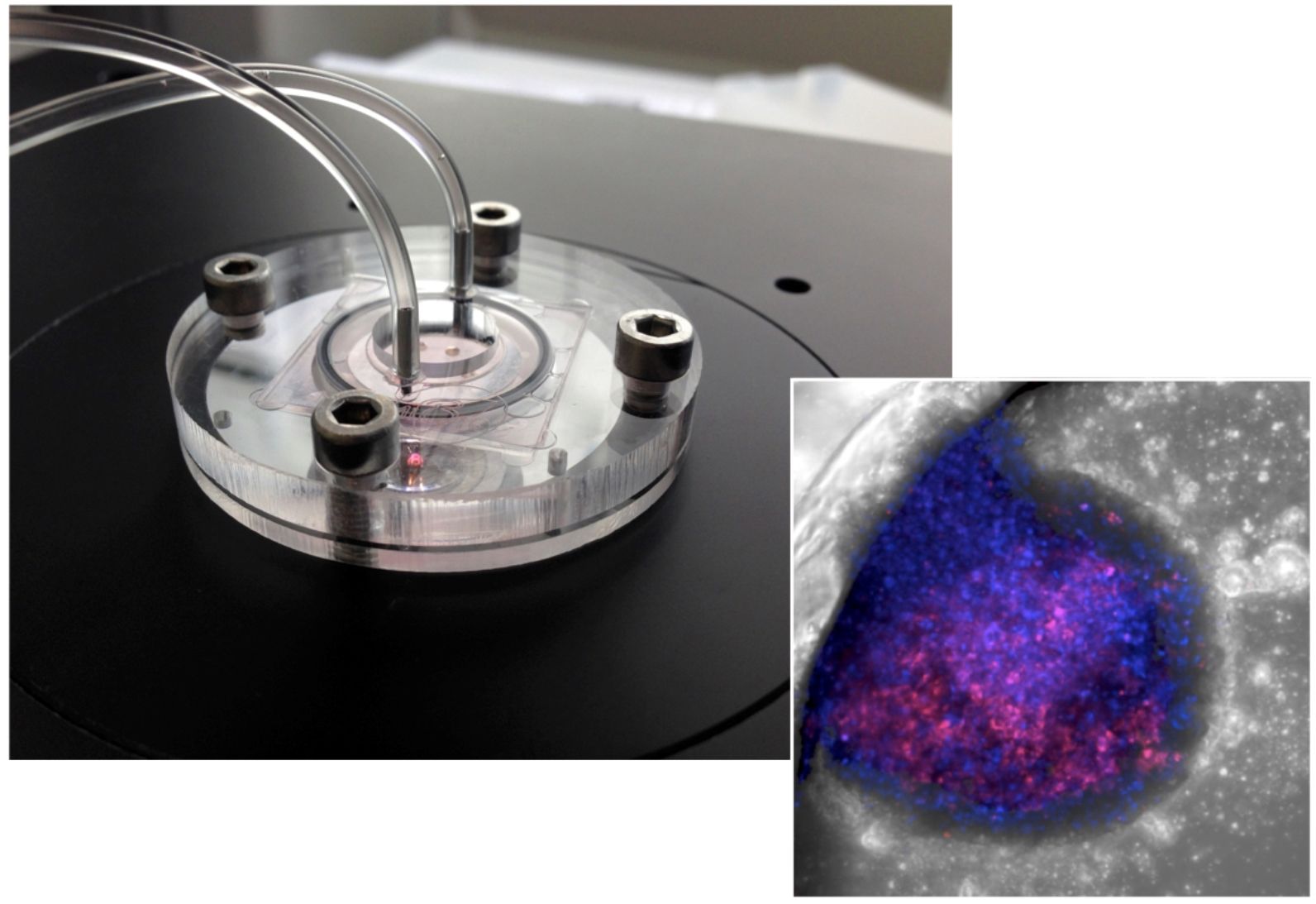
“The liver organs we created were less than a millimeter in diameter and survive for more than a month,” said Prof. Yaakov Nahmias, the study’s lead author and director of Hebrew University’s Alexander Grass Center for Bioengineering.
The breakthrough was due to their idea of adding nanotechnology-based optoelectronic sensors. “We realized that because we are building the organs ourselves, we are not limited to biology, and could introduce electronic and optical sensors to the tissue itself. Essentially we are building bionic organs on a chip,” said Nahmias.
Toxic effects of Tylenol
The sensors in the device allowed them to detect small, rapid changes in cellular respiration that nobody has ever seen before. This enabled them to identify a previously unknown mechanism of toxicity in the common pain reliever acetaminophen (Tylenol), as they reported in the journal Archives of Toxicology.
The finding itself is just as groundbreaking as the bionic liver. Scientists had long believed that acetaminophen broke down in the body into a toxic compound, NAPQI, which could damage cellular respiration. Since the liver naturally deactivates NAPQI, damage was thought to occur only at high doses and in cases of diseased or compromised liver function.
But Nahmias and his team discovered something startling: acetaminophen itself can stop cellular respiration in minutes, even in the absence of NAPQI, much faster and at a much lower dose than previously thought. Thanks to the bionic liver, they now have a likely explanation for off-target effects of acetaminophen.
“This is a fascinating study,” commented Prof. Oren Shibolet, head of the liver unit at Tel-Aviv Sourasky Medical Center, who was not involved in the study. “We knew that acetaminophen can cause nephrotoxicity as well as rare but serious skin reactions, but up until now, we didn’t really understand the mechanism of such an effect. This new technology provides exceptional insight into drug toxicity, and could in fact transform current practice.”
Yissum, the R&D arm of Hebrew University, together with the Fraunhofer Institute, submitted a joint provisional patent application earlier this year and are actively seeking industrial partners to commercialize the liver-on-chip. The global market for this technology is estimated to be worth $17 billion by 2018.
Yissum is also seeking a commercial partner for a human pluripotent stem cell platform developed at the university by Prof. Nissim Benvenisty as an alternative to animal testing for various human genetic diseases and pharmaceuticals for “orphan diseases.”
“The new disease model system developed by Prof. Benvenisty offers a complementary approach that can address many of the hurdles inherent to animal models,” said Yissum CEO Yaccov Michlin. “We are certain that this novel method will have a significant impact on drug screening and development, and Yissum is currently looking for partners in order to further advance this promising approach.”
















