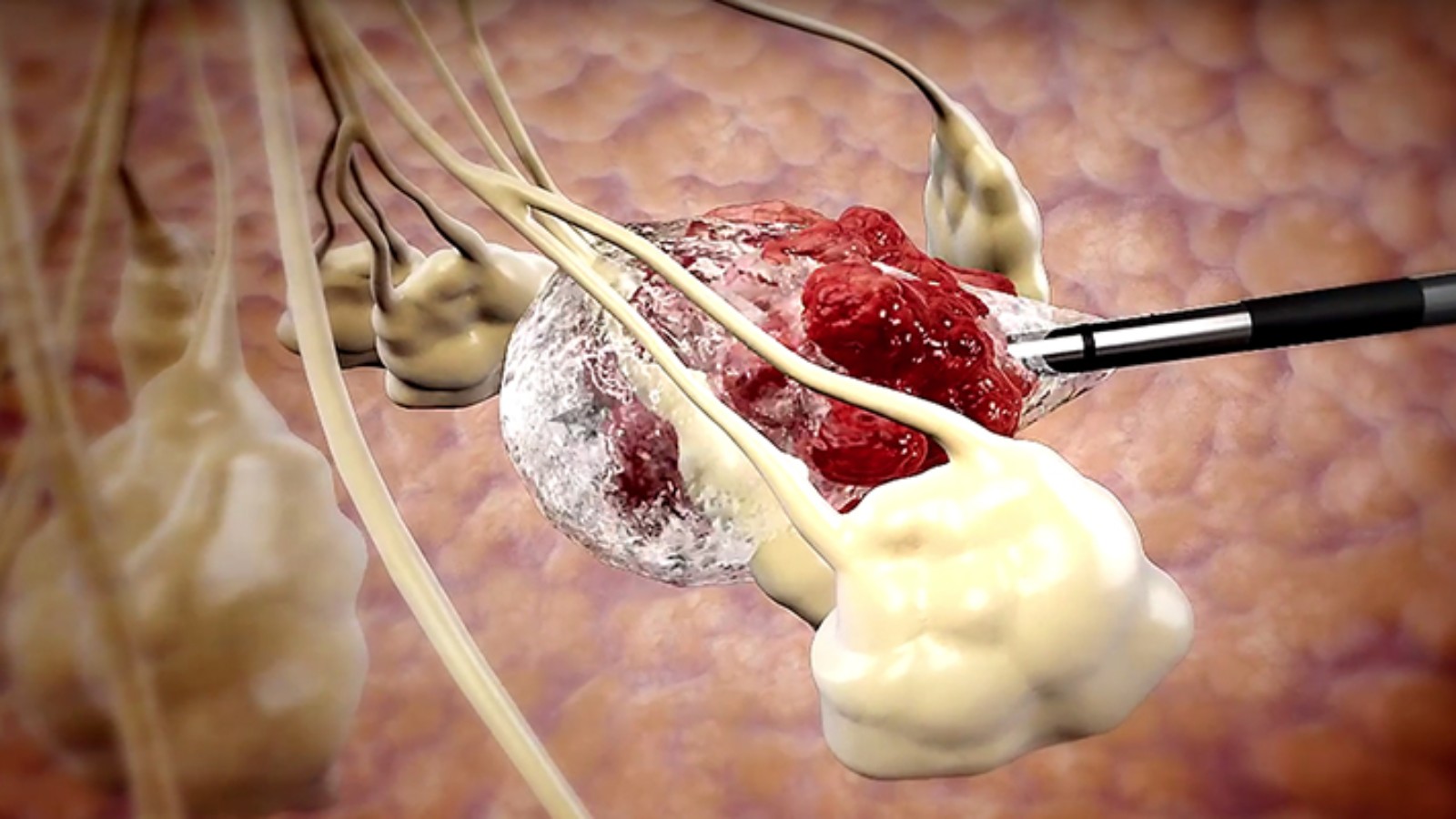
A non-surgical liquid nitrogen (LN2) cryoablation (freezing) technology to destroy benign and malignant tumors demonstrated 100 percent success in a trial with seven kidney cancer patients whose surgically removed tumors had reoccurred.
The outcome of the study, reported in Journal of Molecular and Clinical Medicine, is another feather in the cap of Israeli company IceCure Medical, maker of the ProSense cryoablation system.
The company’s primary focus is breast, kidney and lung cancer. The minimally invasive technology is an alternative to surgical tumor removal and is easily performed in a relatively short outpatient procedure, guided by computerized tomography.
Additionally, IceCure’s solution can help institutions free up operating rooms, while potentially preventing or reducing the risk of infectious pathogens from medical procedures. These are especially critical points during the Covid-19 pandemic.
ProSense is available worldwide after receiving FDA and CE approvals. “Over the past weeks we have completed five successful remote ProSense installations, at clinics in South Africa, Germany, Singapore, Spain, and Thailand,” the company reports.
In the kidney tumor study at Bnai Zion Medical Center in Haifa, the patients’ tumors were destroyed and showed no recurrence in a period of 20 to 36 months following the procedure. The study was done by urologists and radiologists including Sagi Shprits, Robert Sachner, Simona Croitoru, Karina Dorfman, Ofir Avitan, Zaher Bahouth, Amnon Zisman and Ofer Nativ.
In August, IceCure completed an oversubscribed $6 million equity offering. CEO Eyal Shamir said, “The offering ensures the realization of our strategic plan for continued expansion and growth in our operational activities in the US, Europe and Asia.”
Fighting for Israel's truth
We cover what makes life in Israel so special — it's people. A non-profit organization, ISRAEL21c's team of journalists are committed to telling stories that humanize Israelis and show their positive impact on our world. You can bring these stories to life by making a donation of $6/month.