RedHill Biopharma of Tel Aviv is collaborating with two pharmaceutical manufacturers in Europe and Canada to ramp up manufacturing of its drug candidate opaganib (Yeliva) or severe Covid-19 pneumonia.
The orally administered drug, which has anti-inflammatory and antiviral capabilities, could then meet potential demand for emergency use as the pandemic continues worldwide.
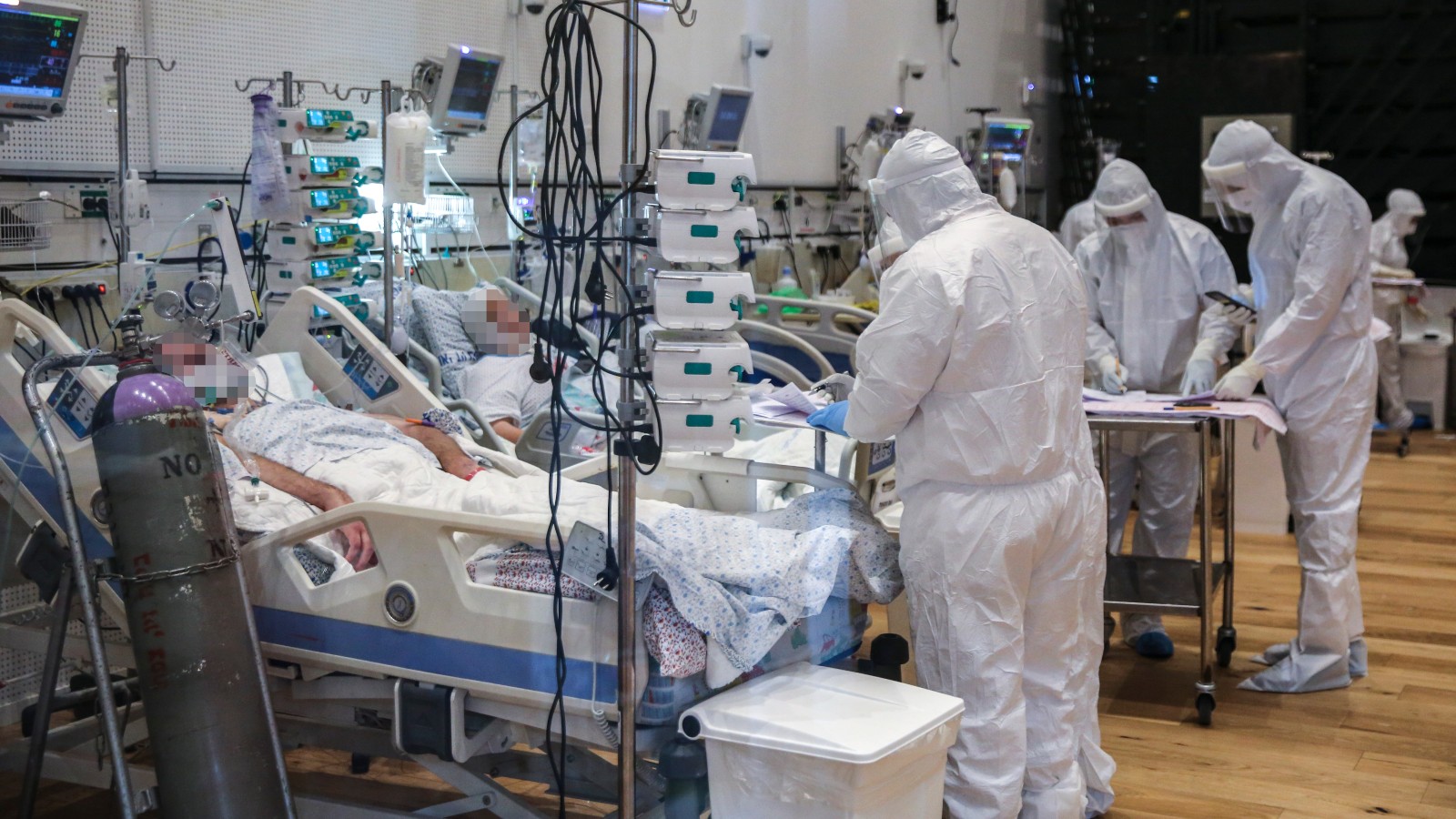
Under a compassionate use program, Covid-19 patients treated with opaganib in an Israeli hospital were discharged without requiring intubation and mechanical ventilation, whereas one-third of the matched case-control group required intubation and mechanical ventilation.
“Opaganib demonstrated potent antiviral activity against SARS-CoV-2, the virus that causes Covid-19, completely inhibiting viral replication in an in vitro model of human lung bronchial tissue,” RedHill reports.

Opaganib is currently in global randomized, double-blind, parallel-arm, placebo-controlled Phase 2/3 and US Phase 2 studies in hospitalized patients with severe Covid-19 pneumonia requiring supplemental oxygen.
The US Phase 2 study is approaching completion of enrollment in eight sites, with initial results expected before the end of this year. The Phase 2/3 study is enrolling up to 270 patients across 15 study sites in six countries by year’s end.
RedHill is in discussions with US government agencies regarding rapid advancement of opaganib manufacturing toward potential emergency use applications.
Opaganib also has the potential to target multiple oncology, viral, inflammatory and gastrointestinal indications. It has FDA orphan drug status for the treatment of cholangiocarcinoma and is being evaluated in a Phase 2a study in advanced cholangiocarcinoma and in a Phase 2 study in prostate cancer.
For the ongoing studies with opaganib, click here