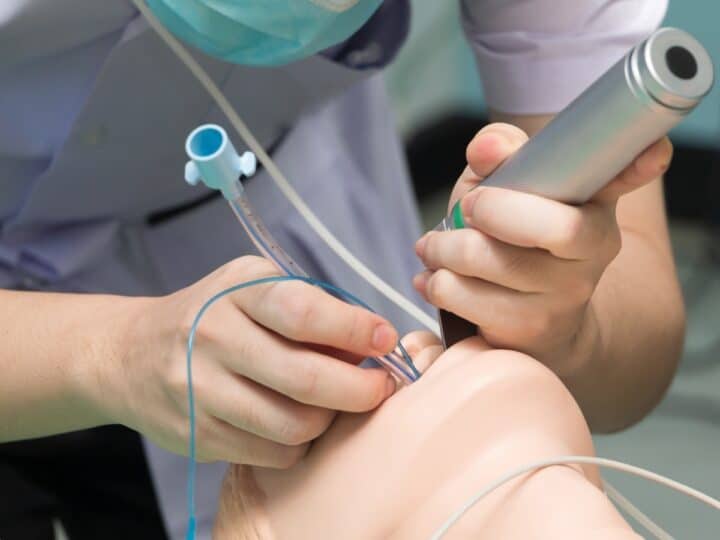
A tiny telescope implant developed in Israel to improve vision in patients with end-stage Age-related Macular Degeneration (AMD) made Popular Science magazine’s annual list of 100 “Best of What’s New” innovations this month.
Israel’s VisionCare Ophthalmic Technologies implantable pea-sized telescope received approval by the US Food and Drug Administration in July for use in a new trademarked patient care program, CentraSight, for patients with visual impairment in both eyes due to untreatable advanced AMD. This retina condition is the most common cause of blindness in the elderly in developed countries.
The first and only treatment for end-stage AMD, VisionCare’s implant is the third-generation version of a device invented in the late 1990s by the company’s Israeli founders, Dr. Isaac Lipshitz and Yossi Gross. The firm is headquartered in California and maintains an R&D, manufacturing and quality assurance facility in Petah Tikva, near Tel Aviv.
“This technology holds significant promise for improving vision and quality of life for individuals with end-stage AMD,” Eli Aharoni tells ISRAEL21c. Aharoni is general manager and vice president of R&D, having joined VisionCare at its inception after heading up one of the Israel military industry’s leading R&D centers. “The new device provides a practical solution to a common problem which currently has no other surgical alternative.”
Pivotal FDA trial
Kathryn A. Colby, an ophthalmic surgeon at Massachusetts Eye and Ear Infirmary in Boston and assistant professor of ophthalmology at Harvard Medical School, calls the device “a breakthrough technology.”
“The clinical results from the pivotal FDA trial have proven we can place this tiny telescope prosthesis inside the eye to help patients see better and, for some, even to levels at which they can recognize people and facial expressions that they could not before,” says Colby.

The miniature telescope is implanted in one eye, enlarging central vision images over a wide area of the AMD-damaged retina, while the non-operated eye provides peripheral vision for mobility and orientation. During rehab, patients are taught how to make the most of their new visual capabilities.
The implant is to be introduced in the United States as part of CentraSight’s four-stage patient management process, Aharoni tells ISRAEL21c.
“First, the patient goes to a retina specialist for an examination and diagnosis. Then, a low-vision expert does an assessment to determine if the patient is a good candidate for the procedure. Implantation is performed as an outpatient procedure by a cornea or cataract surgeon, followed up with rehabilitation by optometrists and occupational therapists.”
Gains in visual acuity and quality of life
VisionCare expects to start training its first teams of CentraSight caregivers in the United States in the near future, once final details are in place for medical insurance reimbursement for the costly procedure. Meanwhile, clinical trials have begun in the United Kingdom, the next likely potential market for the device.
Earlier results from the two clinical trials, conducted at 27 leading ophthalmic centers, were published in scientific journals including Ophthalmology, American Journal of Ophthalmology, and Archives of Ophthalmology. The pivotal clinical trial shows that patients achieved clinically meaningful gains in visual acuity and quality of life with the telescope implant.
Popular Science chose the device as its winner in the health category of “Best of What’s New” in its December 2010 issue after evaluating companies and products in 11 different categories to identify “innovations with potential to positively impact the future and push the envelope of what was not thought to be possible in the previous 12 months.”
Allen W. Hill, CEO of the privately held VisionCare, notes that the advancement of the device from the drawing board to the operating room represents a joint effort among employees, clinical investigators and venture capital investors including Saints Capital, Pitango Venture Capital, Three Arch Partners, Onset Ventures, Infinity Private Equity, Shrem Fudim Group, Vintage Investment Partners and Dovrat.